Packaging
- Supplied as 10 mL single-dose vials
- Bar-coded for inventory management
- Latex-free, Preservative-free
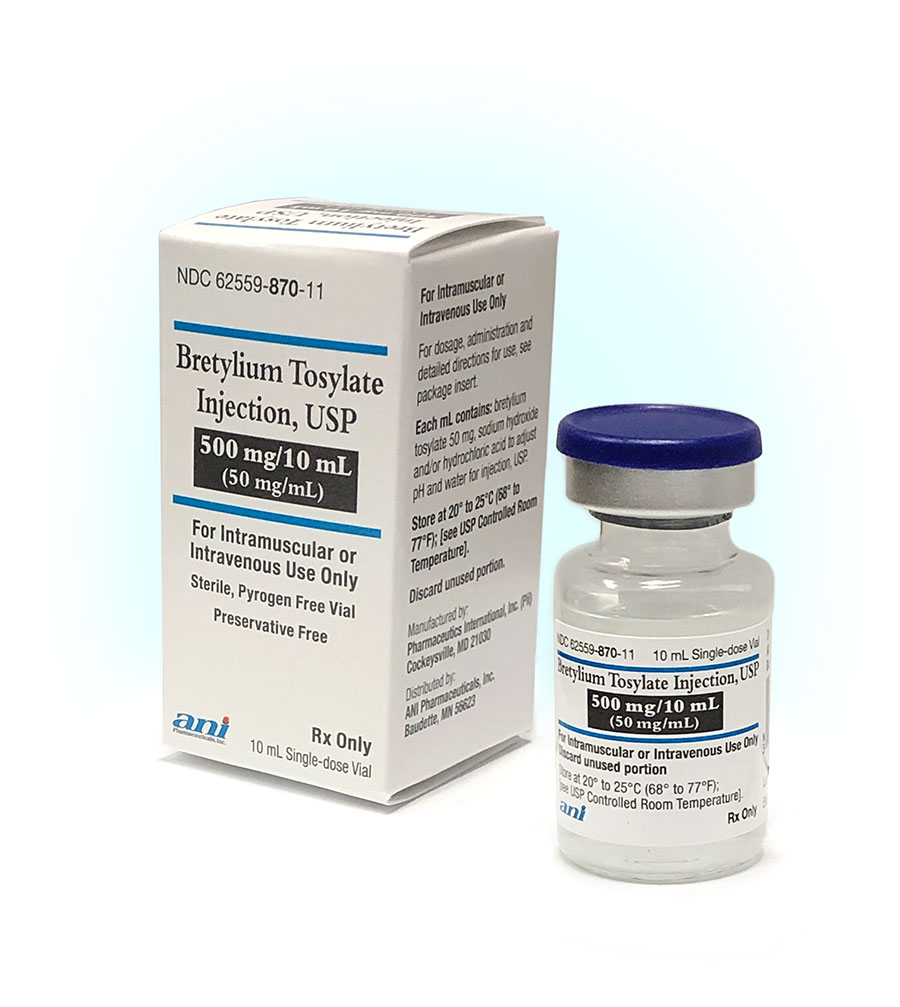
| National Drug Code (NDC): | 62559-870-11 |
|---|---|
| How supplied: | Individual 10 mL single-dose glass vials |
| Store at: | 20° to 25°C (68° to 77°F) |

| National Drug Code (NDC): | 62559-870-11 |
|---|---|
| How supplied: | Individual 10 mL single-dose glass vials |
| Store at: | 20° to 25°C (68° to 77°F) |
Bretylium Tosylate Injection, USP is indicated in the prophylaxis and therapy of ventricular fibrillation.
Bretylium Tosylate Injection, USP is also indicated in the treatment of life-threatening ventricular arrhythmias, such as ventricular tachycardia that have failed to respond to adequate doses of a first-line antiarrhythmic agent, such as lidocaine.
Use of Bretylium Tosylate Injection, USP should be limited to intensive care units, coronary care units or other facilities where equipment and personnel for constant monitoring of cardiac arrhythmias and blood pressure are available.
Following injection of Bretylium Tosylate there may be a delay of 20 minutes to 2 hours in the onset of antiarrhythmic action, although it appears to act within minutes in ventricular fibrillation. The delay in effect appears to be longer after intramuscular than after intravenous injection.
There are no contraindications to use bretylium tosylate in treatment of ventricular fibrillation or life-threatening refractory ventricular arrhythmias, except in the case of digitalis induced arrhythmias. If the arrhythmia is thought to be due to digitalis, bretylium tosylate should not be used.
Hypotension and postural hypotension are the most frequently reported adverse reactions. Nausea and vomiting occurred in about three percent of patients. This is not a complete list of side effects and others may occur. Please see the full Prescribing Information for a complete list of adverse reactions.
To report SUSPECTED ADVERSE REACTIONS, contact ANI Pharmaceuticals, Inc. at 1-800-308-6755 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
This Important Safety Information does not include all the information needed to use Bretylium Tosylate Injection, USP safely and effectively. See full Prescribing Information for Bretylium Tosylate Injection, USP.

You are now leaving bretylium.com
ANI Pharmaceuticals is not responsible for the privacy policy, the content, or the accuracy of any websites accessed through a link on the bretylium.com site. A link to other websites does not constitute an endorsement of ANI Pharmaceuticals or the linked site, its products, or services.
The browser window or tab for bretylium.com will remain open. To return to bretylium.com at any time, close this tab or window.
